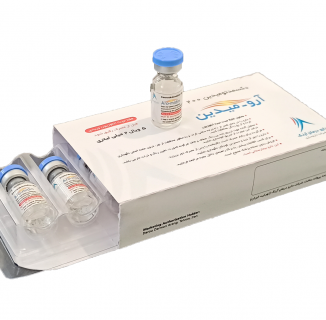
Aro-midin / Dexmedetomidine 200
Dexmedetomidine 200
Aro-midin
Concentrate for solution for infusion
Each vial (2m) contains: Dexmedetomidine (as hydrochloride) 200 micrograms.
Therapeutic indications
- Sedation of initially intubated and mechanically ventilated patients during treatment in an intensive care setting. Dexmedetomidine HCL should be administered by continuous infusion not to exceed 24 hours.
- Sedation of non-intubated patients prior to and/or during surgical and other procedures.
Dosage and Administration
- Dexmedetomidine dosing should be individualized and titrated to desired clinical response.
- Dexmedetomidine is not indicated for infusions lasting longer than 24 hours.
- Dexmedetomidine should be administered using a controlled infusion device.
Dosage Information
|
INDICATION |
DOSAGE AND ADMINISTRATION |
|
Initiation of Intensive Care Unit Sedation |
For adult patients: a loading infusion of one mcg/kg over 10 minutes. For adult patients being converted from alternate sedative therapy: a loading dose may not be required. For patients over 65 years of age: a dose reduction should be considered. For adult patients with impaired hepatic-function: a dose reduction should be considered. |
|
Maintenance of Intensive Care Unit Sedation |
For adult patients: a maintenance infusion of 0.2 to 0.7 mcg/kg/hour. The rate of the maintenance infusion should be adjusted to achieve the desired level of sedation. For patients over 65 years of age: a dose reduction should be considered. For adult patient with impaired hepatic function: a dos reduction should be considered. |
|
Initiation of Procedural Sedation |
For adult patients:a loading infusion of one mcg/kg over 10 minutes. For less invasive procedures such as ophthalmic surgery, a loading infusion of 0.5mcg/kg given over 10 minutes may be suitable. For awakefiberoptic intubation in adult patients: a loading infusion of one mcg/kg over 10 minutes. For patients over 65 years of age: a loading infusion of 0.5 mcg/kg over 10 minutes. For adult patients with impaired hepaticfunction: a dose reduction should be considered.
|
|
Maintenance of Procedural Sedation |
For adult patients: The maintenance infusion is generally initiated at 0.6 mcg/kg/hour and titrated to achieve desired clinical effect with doses ranging from 0.2 to 1 mcg/kg/hour. The rate of the maintenance infusion should be adjusted to achieve the targeted level of sedation. For awakefiberoptic intubation in adult patients:a maintenance infusion of 0.7 mcg/kg/hour is recommended until the endotracheal tube is secured. For patients over 65 years of age:a dose reduction should be considered. For adult patients with impaired hepaticfunction:a dose reduction should be considered. |
Special populations
Elderly
In patients greater than 65 years of age, a higher incidence of bradycardia and hypotension was observed following administration of Dexmedetomidine. Therefore a dose adjustment may need to bo considered in patients over 65 years of age.
Renal impairment
Dexmedetomidine 200 mcg/2 mLinjection pharmacokinetics were not significantly different in patients with severe renal impairment (creatinine clearance: <30 mL/min) compared to healthy subjects.
Hepatic impairment
Since Dexmedetomidineclearance decreases with increasing severity of hepatic impairment, dose reduction may need to be considered in patients with impaired hepatic function.
Pediatric population
Safety and efficacy have not been established for Procedural or ICU Sedation in pediatric patients.
Method of administration
- Dexmedetomidine must be administered only as a diluted intravenous infusion using a controlled infusion device.
- Dexmedetomidine should be administered by healthcare professionals skilled in the management of patients requiring intensive care or in the anesthetic management of patients in the operating room or during diagnostic procedures.
Preparation of solution
Visually inspect parenteral products for particulate matter and discoloration prior to administration whenever solution and container permit.
Dexmedetomidine has the potential to adhere to some types of natural rubber; it is advisable to use administration components made with synthetic or coated natural rubber gaskets.
Dexmedetomidine must be diluted with 0.9% sodium chloride solution to achieve required concentration (4 mcg/mL) prior to administration. Preparation of solutions is the same, whether for the loading dose or maintenance infusion.
To prepare the infusion, withdraw 2 mL of Dexmedetomidine Injection Concentrate, and add to 48 mL of 0.9% sodium chloride injection to a total of 50 mL. Shake gently to mix well.
Administration with Other Fluids
Dexmedetomidine infusion should not be co-administered through the same intravenous catheter with blood or plasma because physical compatibility has not been established.
Dexmedetomidine has been shown to be incompatible when administered with the following drugs: amphotericin B, diazepam.
Dexmedetomidine has been shown to be compatible when administered with the following intravenous fluids:
- 0.9% sodium chloride in water
- 5% dextrose in water
- 20% mannitol
- Lactated Ringer’s solution
-The product should be used immediately and any unused portion should be discarded.
Contraindications
None
Warnings and precautions
Drug Administration
Dexmedetomidineshould be administered only by persons skilled in the management of patients in the intensive care or operating room setting. Due to the known pharmacological effects ofDexmedetomidine, patients should be continuously monitored while receiving Dexmedetomidine.
General precautions
Dexmedetomidine should not be given as a bolus dose and in the ICU a loading dose is not recommended. Users should, therefore, be ready to use an alternative sedative for acute control of agitation or during procedures, especially during the first few hours of treatment. During procedural sedation, a small bolus of another sedative may be used if a rapid increase in sedation level is required.
Caution should be exercised when administering dexmedetomidine to elderly patients. Elderly patients over 65 years of age may be more prone to hypotension with the administration of dexmedetomidine, including a loading dose, for procedures. A dose reduction should be considered
Patient Information: When Dexmedetomidine is infused for more than 6 hours, patients should be informed to report nervousness, agitation and headache that may occur for up to 48 hours.
Hypotension, Bradycardia, and Sinus Arrest
Clinically significant episodes of bradycardia and sinus arrest have been reported with Dexmedetomidineadministration in young, healthy adult volunteers with high vagal tone or with different routes of administration including rapid intravenous or bolus administration.
Reports of hypotension and bradycardia have been associated with Dexmedetomidineinfusion. If medical intervention is required, treatment may include decreasing or stopping the infusion of Dexmedetomidine, increasing the rate of intravenous fluid administration, elevation of the lower extremities, and use of pressor agents. Because Dexmedetomidinehas the potential to augment bradycardia induced by vagal stimuli, clinicians should be prepared to intervene. The intravenous administration of anticholinergic agents (e.g., glycopyrrolate, atropine) should be considered to modify vagal tone. In clinical trials, glycopyrrolate or atropine were effective in the treatment of most episodes of Dexmedetomidine-induced bradycardia. However, in some patients with significant cardiovascular dysfunction, more advanced resuscitative measures were required.
Caution should be exercised when administering Dexmedetomidineto patients with advanced heart block and/or severe ventricular dysfunction. Because Dexmedetomidinedecreases sympathetic nervous system activity, hypotension and/or bradycardia may be expected to be more pronounced in patients with hypovolemia, diabetes mellitus, or chronic hypertension and in elderly patients.
In clinical trials where other vasodilators or negative chronotropic agents were co-administered with Dexmedetomidinean additive pharmacodynamic effect was not observed. Nonetheless, caution should be used when such agents are administered concomitantly with Dexmedetomidine.
Transient Hypertension
Transient hypertension has been observed primarily during the loading dose in association with the initial peripheral vasoconstrictive effects of Dexmedetomidine. Treatment of the transient hypertension has generally not been necessary, although reduction of the loading infusion rate may be desirable.
Patients with hepatic impairment
Care should be taken in severe hepatic impairment as excessive dosing may increase the risk of adverse reactions, over-sedation or prolonged effect as a result of reduced dexmedetomidine clearance.
Patients with neurological disorders
Experience of dexmedetomidine in severe neurological disorders such as head injury and after neurosurgery is limited and it should be used with caution here, especially if deep sedation is required. Dexmedetomidine may reduce cerebral blood flow and intracranial pressure and this should be considered when selecting therapy.
Other
Alpha-2 agonists have rarely been associated with withdrawal reactions when stopped abruptly after prolonged use. This possibility should be considered if the patient develops agitation and hypertension shortly after stopping dexmedetomidine.
Dexmedetomidine may induce hyperthermia that may be resistant to traditional cooling methods. Dexmedetomidine treatment should be discontinued in the event of a sustained unexplained fever and is not recommended for use in malignant hyperthermia-sensitive patients.
Advere Reactions
Intensive Care Unit Sedation
The most frequently reported adverse reactions with dexmedetomidine in the ICU setting are hypotension, hypertension and bradycardia, occurring in approximately 25%, 15% and 13% of patients respectively.
Hypotension and bradycardia were also the most frequent dexmedetomidine-related serious adverse reactions occurring in 1.7% and 0.9% of randomized Intensive Care Unit (ICU) patients respectively.
Procedural sedation
The most frequently reported adverse reactions with dexmedetomidine in procedural sedation are listed below:
- Hypotension (55 % in dexmedetomidine-group vs. 30 % in placebo-group receiving rescue midazolam and fentanyl)
- Respiratory depression ( 38 % in dexmedetomidine-group vs. 35 % in placebo-group receiving rescue midazolam and fentanyl)
- Bradycardia (14 % in dexmedetomidine-group vs. 4 % in placebo-group receiving rescue midazolam and fentanyl)
In relatively healthy non-ICU subjects treated with dexmedetomidine, bradycardia has occasionally led to sinus arrest or pause. The symptoms responded to leg raising and anticholinergics such as atropine or glycopyrrolate. In isolated cases, bradycardia has progressed to periods of asystole in patients with pre-existing bradycardia. Also, cases of cardiac arrest, often preceded by bradycardia or atrioventricular block, have been reported.
Hypertension has been associated with the use of a loading dose and this reaction can be reduced by avoiding such a loading dose or reducing the infusion rate or size of the loading dose.
Pediatric population
Children > 1 month post-natal, predominantly post-operative, have been evaluated for treatment up to 24 hours in the ICU and demonstrated a similar safety profile as in adults. Data in new-born infants (28 – 44 weeks gestation) is very limited and restricted to maintenance doses ≤ 0.2 mcg/kg/h. A single case of hypothermic bradycardia in a neonate has been reported in the literature.
Drug interactions
Co-administration of dexmedetomidine with anesthetics, sedatives, hypnotics, and opioids is likely to lead to an enhancement of effects, including sedative, anesthetic, and cardiorespiratory effects. Specific studies have confirmed enhanced effects with isoflurane, propofol, alfentanil, and midazolam.
No pharmacokinetic interactions between dexmedetomidine and isoflurane, propofol, alfentanil, and midazolam have been demonstrated. However, due to possible pharmacodynamic interactions, when co-administered with dexmedetomidine, a reduction in dosage of dexmedetomidine or the concomitant anesthetic, sedative, hypnotic or opioid may be required.
Fertility, pregnancy, and lactation
Pregnancy
Pregnancy Category C
There are limited amount of data from the use of dexmedetomidine in pregnant women. Studies in animals may have shown reproductive toxicity.
Breastfeeding
Dexmedetomidine is excreted in human milk, however, levels will be below the limit of detection by 24 hours following treatment discontinuation. A risk to infants cannot be excluded. A decision must be made whether to discontinue breastfeeding or to discontinue dexmedetomidine therapy taking into account the benefit of breastfeeding for the child and the benefit of therapy for the woman.
Fertility
In the rat fertility study, dexmedetomidinedid not affect male or female fertility. No human data on fertility are available.
Effects on ability to drive and use machines
Patients should be advised to refrain from driving or other hazardous tasks for a suitable period after receiving Dexmedetomidine for procedural sedation.
Overdose
The tolerability of Dexmedetomidinewas studied in one study in which healthy adult subjects were administered doses at and above the recommended dose of 0.2 to 0.7 mcg/kg/hr. The maximum blood concentration achieved in this study was approximately 13 times the upper boundary of the therapeutic range. The most notable effects observed in two subjects who achieved the highest doses were first degree atrioventricular block and second degree heart block. No hemodynamic compromise was noted with the atrioventricular block and the heart block resolved spontaneously within one minute.
Five adult patients received an overdose of Dexmedetomidinein the intensive care unit sedation studies. Two of these patients had no symptoms reported; one patient received a 2 mcg/kg loading dose over 10 minutes (twice the recommended loading dose) and one patient received a maintenance infusion of 0.8 mcg/kg/hr. Two other patients who received a 2 mcg/kg loading dose over 10 minutes, experienced bradycardia and/or hypotension. One patient who received a loading bolus dose of undiluted Precedex (19.4 mcg/kg), had cardiac arrest from which he was successfully resuscitated.
How supplied
Aro-midin200 is Dexmedetomidine HCL 200 mcg/2mL, Concentrate for solution for infusion. It is supplied in pack of 5 vials of 2 mL.
Storage
Keep in original package and store below 30˚C and protect from light and freezing.
For single use only.Discard any unused solution.
Keep out of the reach and sight of children.
References
PDR 2020
Marketing Authorization Holder: DarouDarmanArang